[Why] is furosemide susceptible to malabsorption from “gut edema”?
Much of medicine is taught using clinical pearls. These small bits of free-standing, clinically relevant information are often based on experience or observation. Others have a backing in pathophysiology, even if the mechanism isn’t known to the teacher or the learners. On the podcast, we’ve covered a number of clinical pearls, including that cancer is a hypercoagulable state, azithromycin is anti-inflammatory, and that calcium stabilizes the membrane in hyperkalemia. Each of these pearls is true, even if most of us are unable to fully explain why. But other clinical pearls don’t have a good physiologic underpinning.
One example: “furosemide is poorly absorbed in decompensated heart as a result of gut edema.”
Part of this pearl is accurate. It is true that furosemide can be poorly absorbed. We often talk about absorption as it relates to oral bioavailability. Furosemide’s oral bioavailability is highly variable, ranging from 10-100%. This variability is both between patients and within the same patient. So if someone is caring for two patients, the first might have really high oral bioavailability, while the other person’s might be very low. But it is also the case that an individual patient might experience periods of high and low bioavailability.
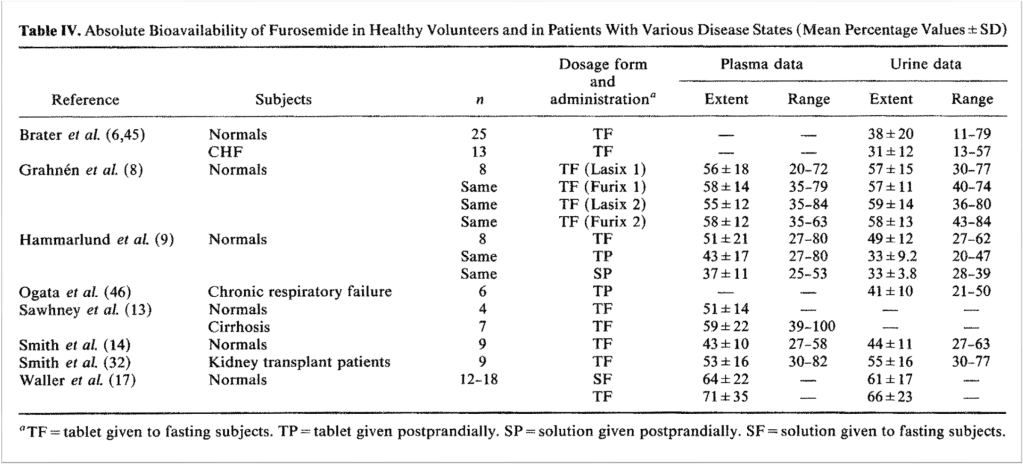
To understand why this matters in heart failure, recall that furosemide is absorbed both in the stomach and proximal small bowel. There is some data suggesting greater absorption in the duodenum than in the stomach.
There are a number of factors that play a role in furosemide’s variable absorption. One very important factor is concomitant food ingestion. There is good data that the absorption of furosemide is negatively affected by food intake, meaning that less is absorbed when it is taken with a meal. And a lot of patients take their medications with meals, so one can see why this might be relevant. This is one of the many reasons why some prefer torsemide; it is unaffected by food intake.
Turning to heart failure, there are 3 key studies comparing furosemide absorption and bioavailability in patients with compensated and decompensated heart failure. The first was published in 1985 by Vasko et al. It included just 11 patients with heart failure. The authors gave these patients oral furosemide during decompensation and again when they were compensated. When compensated, furosemide absorption was faster, and peak plasma concentrations were higher. But, the study also found no significant difference in the area under the curve for plasma concentration when decompensated. Basically, the total amount of furosemide absorbed was the same whether you were compensated or decompensated.
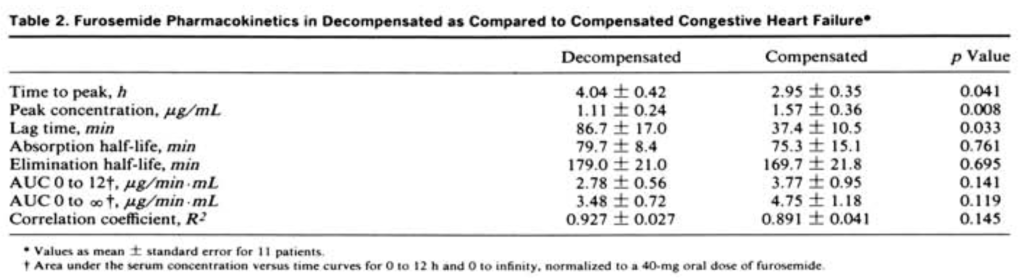
A later study confirmed that there is little difference in total absorption of furosemide between compensated and decompensated states. In fact, this second study found no difference in speed of absorption or peak plasma concentration either. It was the largest of the 3, although it still only included 44 patients.
The third study had similar results. Comparing the decompensated state with the compensated state, they found no differences in the time to peak serum concentration, peak serum concentration itself, or the total amount of furosemide absorbed.
To sum up, three small studies found that the total amount of furosemide absorbed doesn’t decrease, even during decompensation of heart failure. One of the studies did show delayed absorption and decreased peak plasma concentrations, but the other two didn’t confirm this finding.

One very interesting observation made in all 3 studies is that some patients do appear to experience delayed absorption even if, in aggregate, there is no difference. This supports the high variability of furosemide absorption even at the level of an individual patient. Maybe some are more susceptible to the effects of decompensation. If that’s the case, there is no evidence for gut edema leading to any observed differences.
Instead, many propose delayed gastric emptying as the culprit.
There are a few lines of evidence showing that decompensated heart failure is associated with delayed gastric emptying. First, there are experiments in mice demonstrating that the injection of BNP leads to delayed gastric emptying. It also leads to decreased furosemide absorption. Second, left ventricular dysfunction induced by myocardial infarction also leads to both delayed gastric emptying and decreased furosemide absorption. Third, patients with decompensation also have increased sympathetic and decreased parasympathetic tone. This too may lead to delayed gastric emptying.
If the small bowel is the preferred site of furosemide absorption, delayed delivery could be the cause of delayed absorption seen in the Vasko study. And support for this explanation comes from an unexpected source: Roux-en-Y gastric bypass recipients. These patients have rapid gastric emptying. As a result, studies demonstrate they also have a faster time to maximum plasma furosemide concentration and earlier natriuresis. Basically the opposite of decompensated heart failure.
There are other explanations offered for why acute decompensation might lead to a change in furosemide absorption. These include decreased renal blood flow and delivery of furosemide to the nephron and altered blood flow away from sites of absorption. And gut edema does come up in reviews, but no experimental data supporting this explanation could be found.
Despite these findings, our clinical experience supports the idea that patients with decompensated heart failure aren’t responding to oral furosemide. But, a natural experiment that took place a decade ago casts doubt on our observations. In 2012 Ontario faced a shortage of IV furosemide. During this period more oral furosemide was used, even in decompensated heart failure.
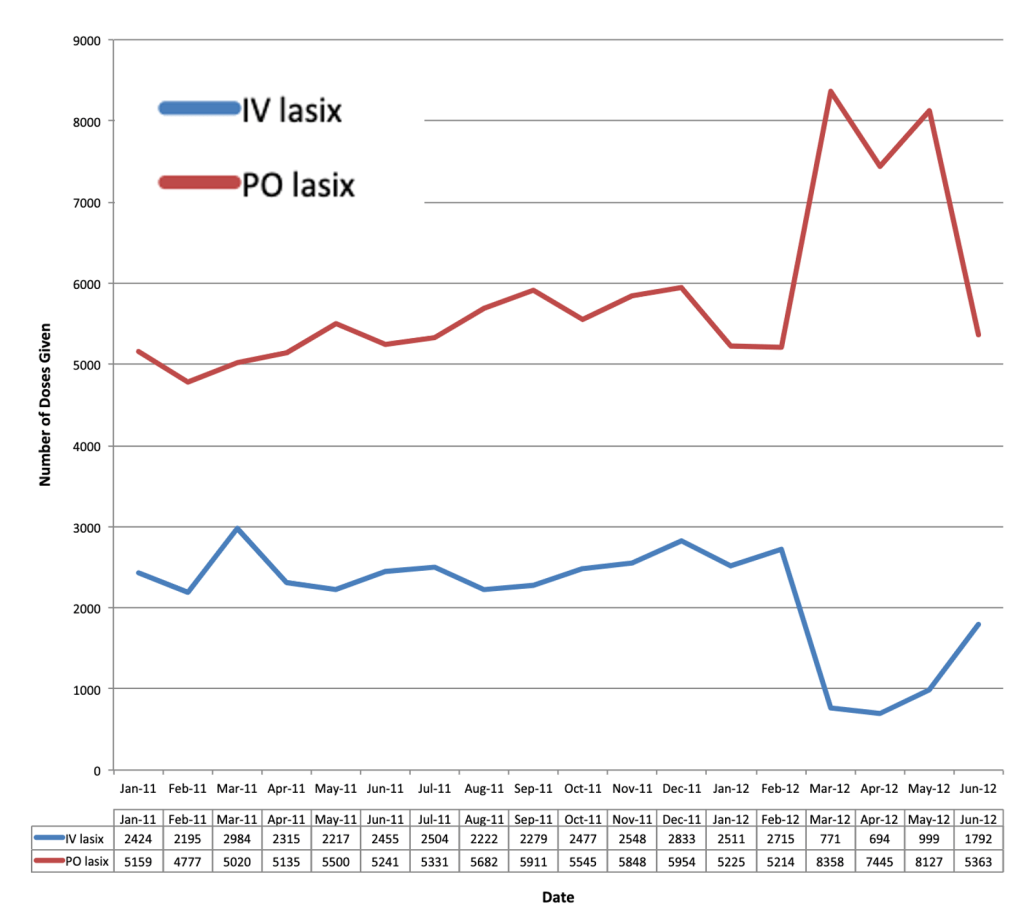
Despite the shift from IV to PO furosemide, hospitals saw no difference in length of stay less than 6 days, ICU admission, 30-day readmissions, or 30-day mortality. They did see a dramatic rise in the use of oral furosemide. This suggests that oral furosemide can work, even in decompensated heart failure.
Given that there is little diuretic or natriuretic effect below a given plasma concentration, known as the threshold, it could be that reduced peak absorption results in less reliable natriuresis. And again, one of the studies showed decreased peak plasma concentration overall. And all three studies offered individual patients for whom this was the case. It could be that the benefit of hospitalization isn’t the use of IV versus PO furosemide as much as it is the use of higher doses. We often see patients go from 20mg of PO furosemide to 40 IV. That’s 4x their home dose. We don’t typically see patients get admitted and placed on 4x their home dose but in oral form. And the study out of Ontario suggests we might be ok with oral furosemide as long as we up the dose enough.
There are other benefits of IV furosemide though. It acts more quickly, making it the standard for acute pulmonary edema. And we just don’t have to worry about the variable bioavailability when we give it IV. As a result, its use in acute heart failure still seems justified.
But, the associated pearl just needs updating. Our suggestion: in decompensated heart failure, furosemide absorption may be delayed as a result of delayed gastric emptying. It has nothing to do with gut edema.
Take Home Points
- Although furosemide absorption may be delayed during decompensated heart failure, the total absorption appears less affected
- Delayed absorption may be related to delayed gastric emptying
- Not all clinical pearls are totally accurate
CME/MOC
Click here to obtain AMA PRA Category 1 Credits™ (0.5 hours), Non-Physician Attendance (0.5 hours), or ABIM MOC Part 2 (0.5 hours).
Listen to the episode
https://directory.libsyn.com/episode/index/id/24550998
Credits & Citation
◾️Episode and show notes by Tony Breu
◾️Audio edited by Clair Morgan of nodderly.com
Breu AC, Abrams HR, Cooper AZ. Gutting a Pearl The Curious Clinicians Podcast. September 28, 2022.
Image credit: https://www.pearlparadise.com/pages/freshwater-information
Related tweetorial: https://mobile.twitter.com/tony_breu/status/1476978129520496643

