Why doesn’t hemolysis cause acute kidney injury as easily as rhabdomyolysis?
Whenever we see a patient with rhabdomyolysis one of the biggest concerns is acute kidney injury (AKI). We reflexively go from looking at the creatinine kinase (CK) to looking at creatinine. But we see a lot of hemolysis and don’t have the same reflex. In fact, many of us cannot recall a single case of hemolysis-mediated AKI.
Both hemoglobin and myoglobin contain heme. And heme is the toxic molecule that causes a form of renal injury called pigment nephropathy. Therefore, conditions that lead to the release of lots of heme-containing molecules, whether it be hemoglobin or myoglobin, should both lead to tubular injury. But that doesn’t seem to be the case. At least not to the same degree.
It is important to establish that hemolysis can lead to AKI. Historically, massive hemolysis from ABO mismatch was a major cause. Now the causes are more varied.

Paroxysmal nocturnal hemoglobinuria is one hemolytic condition often associated with AKI. More than 90% of patients with PNH exhibit hemoglobinuria at some point, with approximately half of the patients presenting with AKI. But this is the exception. Most other hemolytic conditions aren’t big risk factors for AKI.
Cohort studies suggest that the risk of AKI with rhabdomyolysis is somewhere between 10% and 50% with the risk increasing with increasing CK levels. And one study reported a prevalence of biopsy-proven pigment nephropathy of 0.06% for hemolysis versus 0.8% for rhabdomyolysis. Hemolysis was 13 times less likely to cause pigment nephropathy.
The explanation for the difference in these rates starts with the realization that heme must cross the glomerulus and gain access to the renal tubules in order to cause AKI.
The first line of defense comes from haptoglobin, a protein that exists to protect us from the toxic effects of free hemoglobin. Basically, haptoglobin binds free hemoglobin in an attempt to neutralize it. This explains why haptoglobin levels are low in hemolysis. Given the large size of the hemoglobin:hatoglobin complexes, they are not filtered by the kidney and cannot injure the tubules.
Recall that we use a low haptoglobin to diagnose hemolysis. This demonstrates that we exhaust this system quickly. We just don’t have enough haptoglobin to bind all the free hemoglobin in the setting of hemolysis.
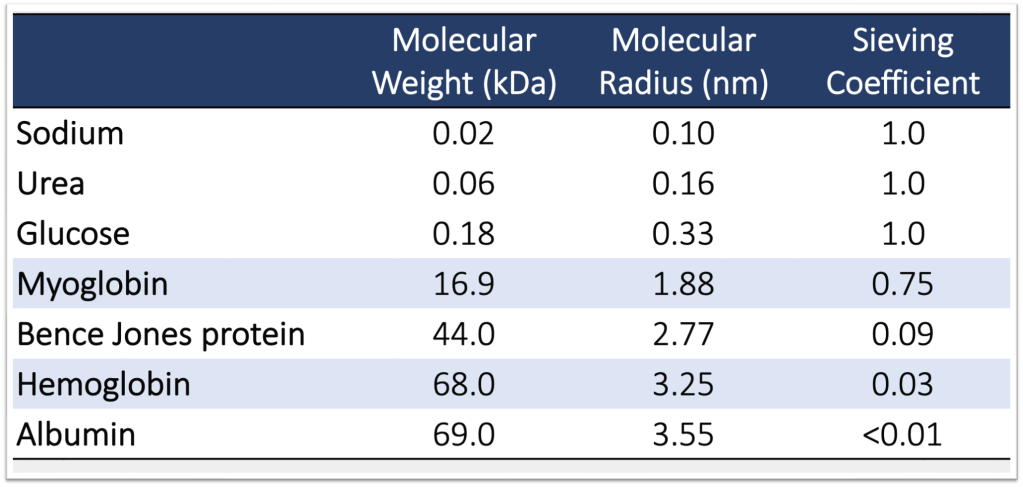
The second mechanism relates to the structure of hemoglobin itself. Hemoglobin is a tetramer containing four globin proteins and has a molecular weight of 68kd. This is nearly identical to albumin which has a molecular weight of 69kd. So it is a modestly-sized molecule. In episode 48 we discussed the sieving coefficient and how it provides insights into how much of a protein is filtered by the glomerulus. We said in that episode that albumin has a sieving coefficient of <0.01 meaning almost no albumin is filtered by the glomerulus.
Hemoglobin, with its similar size, also has a low sieving coefficient of 0.03. This means that only about 3% of hemoglobin traverses the glomerulus. and can access the renal tubule to cause AKI.
Myoglobin is 17 kd. So it is significantly smaller than both hemoglobin and albumin. This translates into a far higher sieving coefficient of 0.75. This means that 75% of the myoglobin delivered to the glomerulus is filtered. This is enormous compared with 3% for hemoglobin.
The question now becomes: how does hemolysis ever cause AKI if so little is filtered by the glomerulus? First, remember that we need massive hemolysis for AKI to result. ABO mismatch is the classic example. In these conditions, even a low sieving coefficient may not protect against large amounts of hemoglobin being filtered.
But there is more to it. It turns out that hemoglobin exists not just as tetramers but also as dimers. These exist in an equilibrium with the tetramers with most existing as the larger tetramers. Unsurprisingly, it is the dimers that undergo more renal excretion as compared with tetramers. In short, it is likely the dimers that get filtered and cause injury. But because the equilibrium favors tetramers there needs to be a lot of hemolysis before enough are around to cause AKI.
Take Home Points
- Even though both myoglobin and hemoglobin containe heme, the toxic molecule in pigment nephropathy, rhabdomyolysis causes more acute kidney injury than hemolysis.
- One reason is that haptoglobin binds hemoglobin, preventing filtration by the glomerulus.
- Myoglobin is a mononer and is easily filtered. Hemloglobin is a tetramer and is not filtered as easily leaving it unavailable to cause tubular injury.
- Some hemoglobin exists as a dimer; these dimers are likely the main form that get filtered and cause AKI in hemolysis
CME/MOC
Click here to obtain AMA PRA Category 1 Credits™ (0.5 hours), Non-Physician Attendance (0.5 hours), or ABIM MOC Part 2 (0.5 hours).
Listen to the episode
https://directory.libsyn.com/episode/index/id/24079833
Credits & Citation
◾️Episode and show notes by Tony Breu
◾️Audio edited by Clair Morgan of nodderly.com
Breu AC, Abrams HR, Cooper AZ. A Cute Pigment The Curious Clinicians Podcast. August 17, 2022.
Image credit: https://www.arkanalabs.com/ati-with-pigmented-casts/
Related tweetorial: https://x.com/tony_breu/status/1378086086044479493?s=20

