Why can cefepime cause neurotoxicity?
Cefepime neurotoxicity is an under-recognized clinical entity and the potential for its presence can confound the assessment of encephalopathic patients. But at the same time, it’s a very effective broad-spectrum antibiotic (i.e., it treats both gram-positive and gram-negative organisms) that clinicians should not fear to use.
As background, cefepime is a 4th generation cephalosporin antibiotic. It is very effective against gram-negative infections, including Pseudomonas. It easily crosses the blood-brain barrier making it a good option for the treatment of meningitis. Dosing can be tricky in patients with renal failure as it is excreted exclusively by the kidney.
The first report of neurotoxicity associated with cefepime occurred relatively recently, back in 1999. It was published as a letter in the journal Nephrology Dialysis Transplantation. A patient with end stage renal disease was treated with high doses of cefepime and 4 or 5 days into their course developed confusion, tremors, myoclonic jerks, and tonic-clonic seizures. The clinicians in this case used reverse-phase high performance liquid chromatography to measure serum levels of cefepime. They were, not surprisingly, high. After receiving several dialysis sessions the serum drug levels came down and his neurological symptoms resolved. This strongly suggested that cefepime was the cause of the neurologic manifestations.
Despite the astute work of the clinicians who first reported it, cefepime neurotoxicity can be a difficult diagnosis to make and it is one of those situations where one must have a high index of suspicion. The spectrum of symptoms are broad and include anything from mild encephalopathy or confusion to tremors or myoclonus to seizures. We have the most clinical evidence for an association with seizure risk, perhaps because those events are the easiest to observe.
Timing of drug initiation versus symptom onset is important when considering the diagnosis, as with any drug toxicity. And there actually are EEG patterns that are consistent with a drug-related encephalopathy, such as triphasic waves or non-convulsive status epilepticus. But at the same time, clinicians shouldn’t anchor on this diagnosis either and miss other neurological pathologies.
Renal failure is the main risk factor for cefepime neurotoxicity, particularly when one fails to dose-adjust the drug for GFR. In one cohort study published in the journal Critical Care in 2013, the authors looked at ICU 100 patients who received cefepime. Of the 15 patient who demonstrated some form of neurotoxicity, 13 had acute kidney injury or chronic kidney disease and only 4 had appropriate dose-adjustment for renal function.
The metabolic milieu of renal failure may matter as well. A study in 2018 looked at cefepime’s ability to induce seizures in rat brains. The authors bathed the brains in a hyperkalemic medium and a normokalemic medium, and then exposed both to cefepime. The rat neurons surrounded by higher potassium levels had more seizures after cefepime exposure than the brains surrounded in normal potassium. In patients with renal failure, the risk of cefepime neurotoxicity likely reflects both drug levels and the metabolic milieu around neurons.
The ability for antibiotics to induce neurotoxicity is not unique to cefepime, particularly inducing seizures, even though it may be the most common offender. Although many learn about carbapenem-induced seizures, all beta-lactam antibiotics, including cephalosporins, penicillins, and carbapenems can cause neurotoxicity. It is a class effect, though cefepime and cefazolin seem to be the most likely to cause seizure with piperacillin and cefuroxime less likely.

The explanation for this class-effect is in the name: beta-lactam. All beta-lactam antibiotics have beta-lactam rings as part of their structures. A 1971 study in cats implicated the beta-lactam ring specifically as the source of neurotoxicity with these types of antibiotics. The researchers monitored brain activity while giving the cats doses of penicillin high enough to induce seizures. When they administered the enzyme beta-lactamase, which degrades beta-lactam rings, the cats were protected from getting seizures induced by penicillin. Something about breaking down the beta-lactam ring blocked the ability of penicillin to cause neurotoxicity.
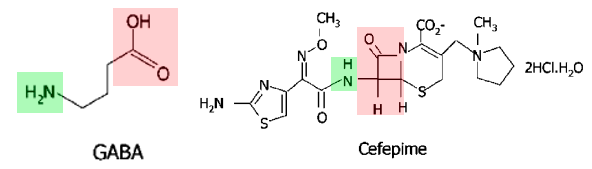
Cephalosporins block the binding of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA) to its receptor in the brain. This was demonstrated with cefazolin in a rats study in which researchers found that the drug physically blocked the GABA receptor. And it did so in a concentration-dependent manner; the more cefazolin was present the more the GABA receptor was blocked. Presumably the same would apply to cefepime.
There are two main types of GABA receptors, appropriately named subtype A and B. Subtype A is a ligand-gated chloride channel. Cefepime binds fairly well to the GABA-A receptor and blocks chloride influx, blocking GABA-A’s ability to respond to GABA signaling. Among the cephalosporins, those that bind GABA-A more tightly are more able to induce seizure activity.
GABA and the beta-lactam ring have structural similarities with each other, which probably explains why beta-lactam antibiotics have the potential to wreak disinhibited havoc in the central nervous system.
Take Home Points
- Cefepime can induce neurotoxicity by blocking the GABA receptor, similar to other beta-lactam antibiotics.
- Neurotoxicity includes seizures, encephalopathy, myoclonus, and tremors.
- Neurotoxicity likely results from structural similarities between GABA and the beta-lactam ring. This is a class effect seen with other beta-lactam antibiotics.
- Renal failure is the main risk factor as it leads to decreased clearance and increased serum drug levels. This results in more blockade of GABA receptors.
- Hyperkalemia likely also predisposes to cefepime-induced neurotoxicity.
CME/MOC
Click here to obtain AMA PRA Category 1 Credits™ (0.5 hours), Non-Physician Attendance (0.5 hours), or ABIM MOC Part 2 (0.5 hours).
Listen to the episode
https://oembed.libsyn.com/embed?item_id=23383166
Credits & Citation
◾️Episode written by Avi Cooper
◾️Show notes written by Avi Cooper and Tony Breu
◾️Audio edited by Clair Morgan of nodderly.com
Breu AC, Cooper AZ, Abrams HR. Antibiotics on the brain. The Curious Clinicians Podcast. June 8, 2022.
Related tweetorial: https://x.com/AvrahamCooperMD/status/1353394052448849920?s=20

