For this week’s episode, we discussed drug names. More specifically, we talked about how one drug’s name says a lot about its development and mechanism of action.
The drug: Sinemet (carbidopa/levodopa).
To understand the origin story of Sinemet, one must first recall that the key pathologic feature of Parkinson disease (the most common indication for Sinemet) is the loss of cells in the substantia nigra, with a resulting decrease in dopamine levels in the brain. The obvious response to this would be to just give these patients dopamine. Unfortunately, dopamine does not cross the blood-brain barrier. As a result, it isn’t a treatment option for Parkinson disease.
Even though dopamine can’t cross the blood-brain barrier, its precursor, dihydroxyphenylalanine can. We, of course, just call this DOPA. Once in the brain, DOPA is converted to dopamine. Making use of this knowledge, George Cotzias, Melvin Van Woert, and Lewis Schiffer gave DOPA to 16 patients with Parkinson disease. Their results were published in 1967 in the NEJM. It was a positive study in that the patients Parkinson symptoms got better. The downside: 4 of 16 patients experienced neutropenia. This clearly would not be an acceptable side effect profile.
We don’t typically associate levodopa – which is DOPA – with neutropenia. So, what explains the high rate of neutropenia? Like many molecules DOPA is chiral and has two enantiomers. In the study by Cotzias, the molecule given contained both the left and right handed enantiomers. Soon after it was discovered that the left-handed enantiomer (L-dopa) is the active form. And, it turns out our body only makes L-dopa. This allowed L-dopa to be administered without the right-handed enantiomer (D-dopa or dextrodopa), which was likely the culprit for the neutropenia. This introduced us to levodopa (the “levo” referring to “levorotation”, the left handed chirality of this isomer).
Why not just use Levodopa monotherapy?
Though progress was being made, the ability to escalate doses to maximize therapeutic benefit was limited by gastrointestinal side effects. For example, one study reported rates of nausea and vomiting of 91% and 55%.
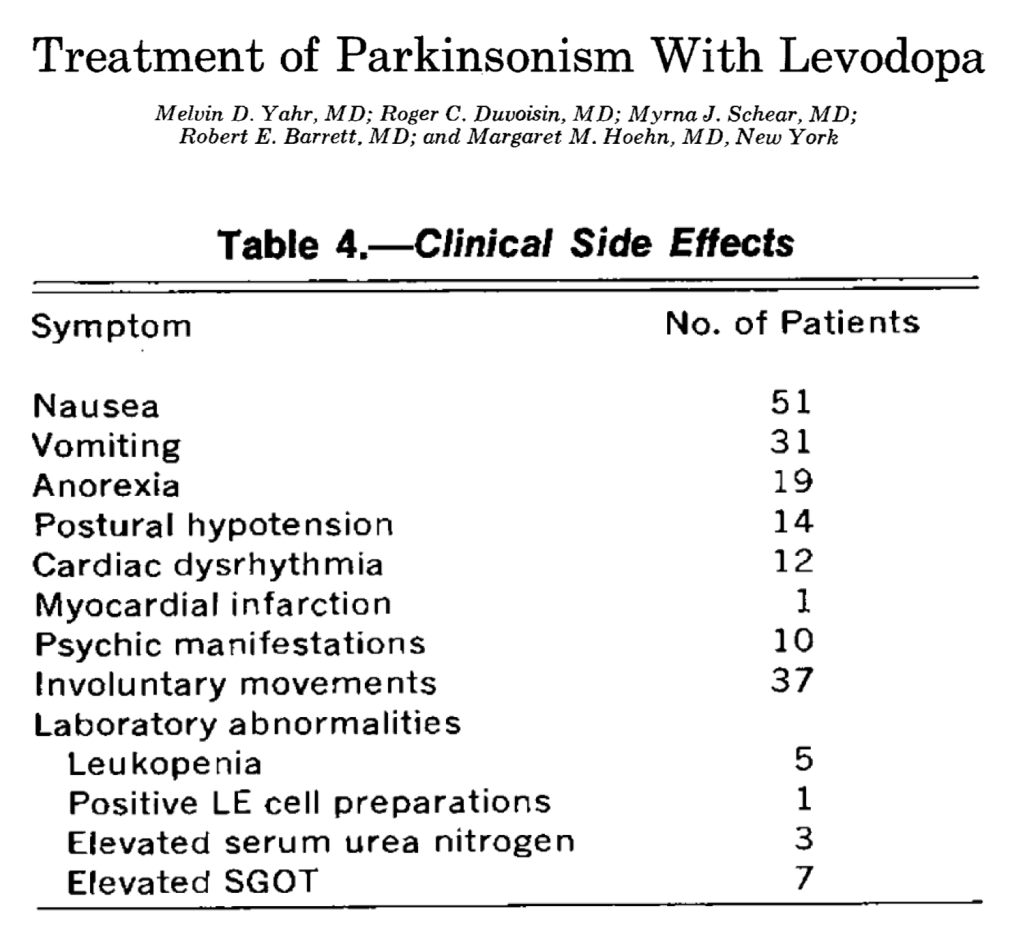
What’s interesting is that levodopa is largely inert. Its effects in Parkinson disease derive from the fact that it crosses the blood-brain barrier and is converted to dopamine in the brain. The problem is that levodopa is also converted to dopamine peripherally. This leads to gastrointestinal side effects via activation of D2 receptors in the area postrema in the medulla.1 A solution to this problem was needed.
Enter: carbidopa
Fortunately, there is a drug that perfectly addresses the issues with levodopa. Carbidopa inhibits DOPA decarboxylase, the enzyme that converts levodopa to dopamine. Trials suggest that if you administer carbidopa with levodopa, the rates of nausea decrease dramatically.
And because carbidopa doesn’t itself cross the blood-brain barrier, it doesn’t inhibit conversion in the brain! The result is that there is very little peripheral conversion of levodopa to dopamine (protecting against nausea and vomiting) and no inhibition of conversion in the brain (the exact place where you want it to take place). Carbidopa really is the perfect molecule to pair with levodopa.
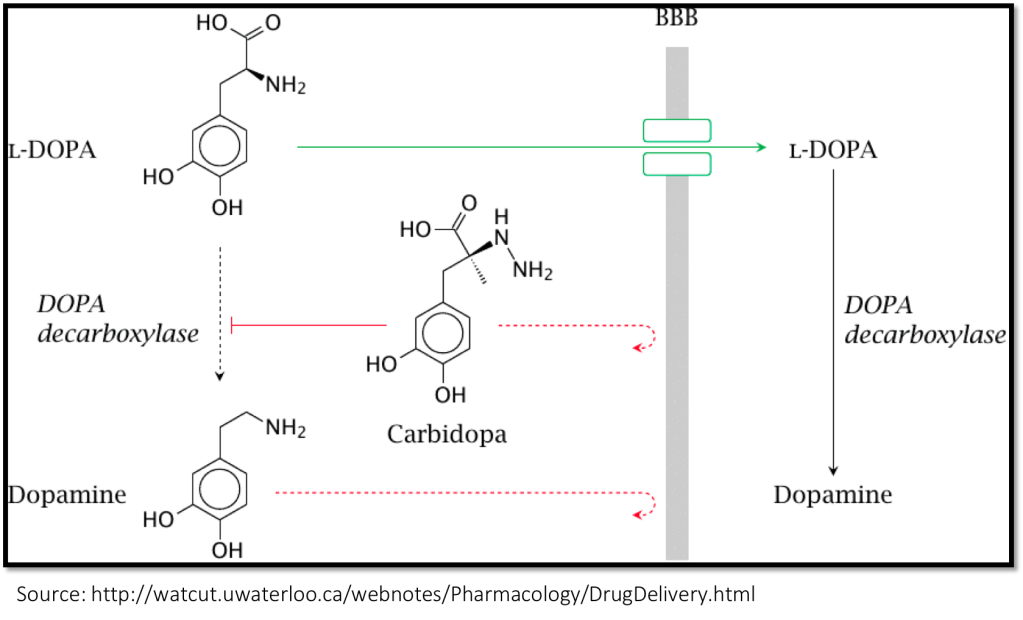
The namers of this new combination medication made reference to the added benefit of carbidopa added to levodopa.
SINE = without
EMET = vomiting
Together, you get SINEMET.
Other great names
In response to a tweetorial on this topic, tons of people added their favorite origin stories. The one most often cited is Lasix being named because it lasts six hours. Some others include:
Those named for their site of discovery/development or their developer:
- Montelukast was named for Montreal, where it was developed.
- Nystatin is apparently named for the NY State Health Department where the research had been done.
- Warfarin is from the (University of) Wisconsin Alumni Research Foundation.
- Rapamycin (sirolimus) is a natural product isolated from Streptomyces hygroscopicus, found on the island of Rapa Nui. There is a great RadioLab episode on this.
- Carfilzomib was developed by molecular biologist (a founder of the company that first developed carfilzomib) Philip Whitcome. The drug was named for him and his wife Carla.
A pair of acid suppressors are well-named:
- Protonix (pantoprazole) “nixes the protons”.
- Prevacid (lansoprazole) prevents acid.
And the list goes on:
- Ambien: AM bien!
- Epinephrine comes from it being “epi” – as in over or on – the neph – as in kidney. Adrenalin comes from it be “ad” – as in near – the renal – as in kidney.
- Vicodin comes from the fact that it is 6 times stronger than codeine so, they used VI (the roman numeral for 6) + CODeINe (dropped the e’s from codeine).
- Tacrolimus is produced by a soil bacterium, Streptomyces tsukubaensis. The name tacrolimus is derived from “Tsukuba macrolide immunosuppressant”.
- Vemurafenib: V to E mutated raf inhibition for BRAF mutated Melanoma or Hairy Cell Leukemia.
- Heparin was initially isolated from dog livers, thus hep.
- Lispro insulin has an amino acid sequence reversal at LYSine and PROline. Hence Lyspro (Lispro) insulin.
- NPH is neutral protamine Hagedorn named for neutral pH protein by Hans Christian Hagedorn (an insulin researcher).
- Glucophage (metformin) helps the body eat glucose.
- Namenda (memantine) was named after its mechanism of action as a NMDA-receptor antagonist.
- Lopressor (metoprolol) lowers blood pressure. Also metoprolol may come from (me-too)prolol, developed to counter the success of propranolol.
- Flomax (tamsulosin) leads to maximum urinary flow.
- Macrobid (nitrofurantoin) is macrodantin dosed BID.
- Premarin derives from pregnant mares urine.
- Augmentin (amoxicillin/clavulanic acid) was named because the clavulanic acid augments the action of the amoxicillin.
- Levofloxacin is the L enantiomer of oflixacin which is a racemic mixture.
Take Home Points
- Parkinson disease results from decreased brain dopamine.
- Levodopa is converted to dopamine in the brain, treating symptoms.
- But, conversion in the periphery also occurs, leading to nausea and vomiting.
- Carbidopa decreases peripheral conversion of levodopa to dopamine.
- This mitigates nausea resulting from activation of D2 receptors in the area postrema.
CME/MOC
CME/MOC credits are not available for this episode.
Listen to the episode
https://oembed.libsyn.com/embed?item_id=20330774
Credits & Citation
◾️Episode and show notes written by Tony Breu
◾️Audio edited by Clair Morgan of nodderly.com
Breu AC, Cooper AZ, Abrams HR. How does Sinemet’s name provide insights into why it was developed and its mechanism of action? The Curious Clinicians Podcast. September 1, 2021
Image credit: https://library.neura.edu.au/schizophrenia/physical-features/functional-changes/biochemical-changes/dopamine/
1I didn’t remember this fact from anatomy, but recall that the area postrema is actually outside the blood-brain barrier and therefore can be reached by peripheral dopamine. In fact, one role of this structure is to sense blood toxins and cause us to vomit.
Related tweetorial: https://x.com/tony_breu/status/1093977481693007872

Edoxaban, a DOAC made by Daiichi-Sankyo, a Japanese phama…named after Edo, the old name of Tokyo.
LikeLike