On this episode of the podcast, the group answered a question that is often addressed with a wave of the hand: How do diuretics lead to metabolic alkalosis? Along the way, we dissected what is meant by contraction alkalosis, introduced a protein that none of us had heard of until exploring this question, and found a new appreciation for an often-unnoticed part of the basic metabolic panel. Here is a link to the tweetorial on this topic that Tony posted on February 15, 2020.
“Contraction Alkalosis” First Appears
After the emergence of potent loop diuretics many began to observe a resulting alkalosis. The idea that this might be the result of ‘contraction’ of the volume around a fixed amount of bicarbonate first appeared in a 1965 study published in the Annals of Internal Medicine.

The study included 4 patients with heart failure. They received ethacrynic acid (a loop diuretic) at a dose of 50mg four times daily. The study participants made a lot of urine (3-7 liters per day for 1-3 days) and developed metabolic alkalosis. Here is a figure demonstrating the change in pH and plasma bicarbonate. You’ll notice that both rise in the experimental group (right side of each figure).
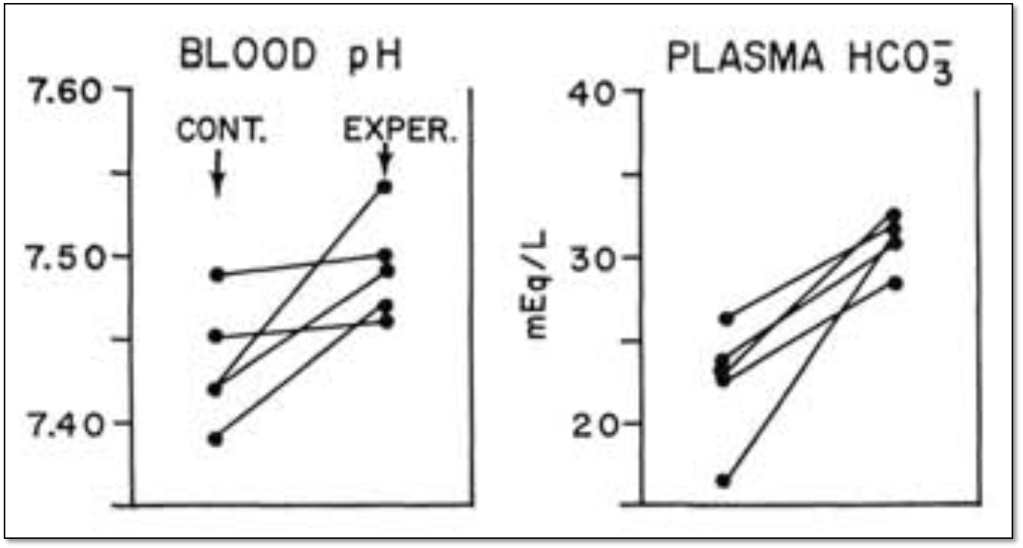
The authors note that “Nearly all of the bicarbonate initially present in the extracellular fluid could be accounted for in the final volume of extracellular fluid.” Because the amount of bicarbonate hadn’t changed, the increase in plasma concentration was attributed to a decrease (i.e., contraction) around this fixed volume. The idea of a “contraction alkalosis” was born.
Generation versus Maintenance
The 1965 study by Cannon et al demonstrated that a metabolic alkalosis might be generated by contraction after diuretic use. But, even if that occurred, something must maintain the metabolic alkalosis.
This distinction is important because the kidneys have a remarkable ability to get rid of excess bicarbonate. In one study, normal subjects were given up to ~1000 mEq of sodium bicarbonate per day for up to 3 weeks. One patient even received 1680 mEq daily.
[Aside #1 to provide context: Assuming a normal serum bicarbonate of 24 mEq/L and a normal plasma volume of approximately 3 liters, the body typically has less than 100 mEq of bicarbonate in the extracellular compartment. Again, these study subjects were taking in about 10 times that amount.]
[Aside #2 to provide (more) context: Sodium bicarbonate tablets typically have either 325mg or 650mg of bicarbonate. This amounts to approximately 4-8 mEq per tablet. So, this study administered the equivalent of ~125-250 sodium bicarbonate tabs each day.]
Here’s the key observation from this study: patients excreted virtually all the excess bicarbonate and developed only a minor increase in plasma bicarbonate concentration. Disorders associated with maintained metabolic alkalosis therefore must involve impairment of the ability to excrete excess bicarbonate.
Why can’t the kidney excrete the excess bicarbonate?
If diuretic-induced contraction cannot maintain a metabolic alkalosis, what other mechanisms are at play. One is the resulting volume depletion, which often results from aggressive diuresis.
- Volume depletion & decreased GFR. As one becomes volume depleted there is a decrease in glomerular filtration rate (GFR). With decreased GFR comes decreased filtered bicarbonate. As the amount of bicarbonate filtered decreases, there is a resulting decrease in the amount excreted.
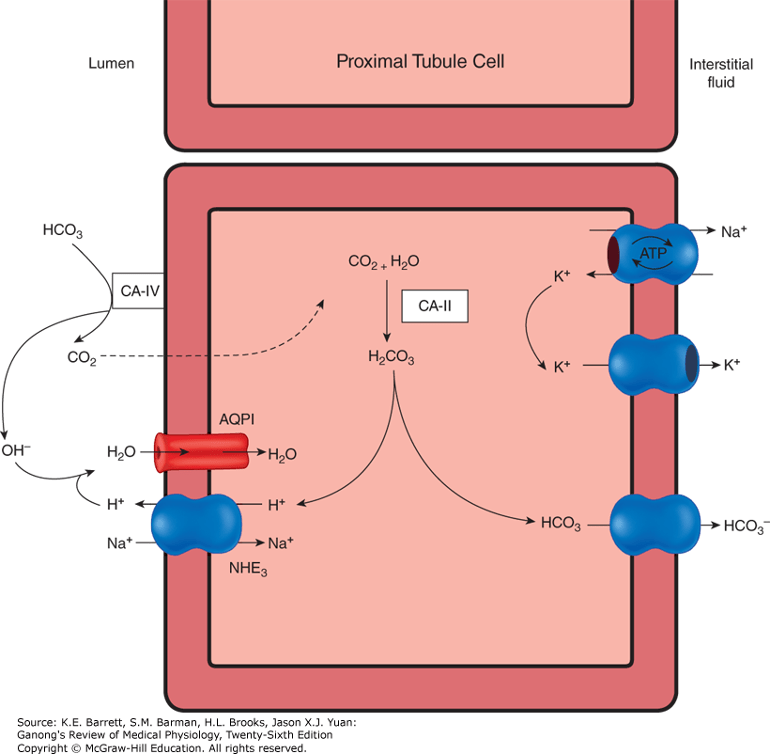
- Volume depletion & increased proximal tubular reabsorption. Volume depletion also leads to an increase in proximal tubule reabsorption of sodium. As a result, there is an increase in activity of the sodium and hydrogen ion antiporter. As hydrogen ions are secreted, there is generation of new bicarbonate via carbonic anhydrase.
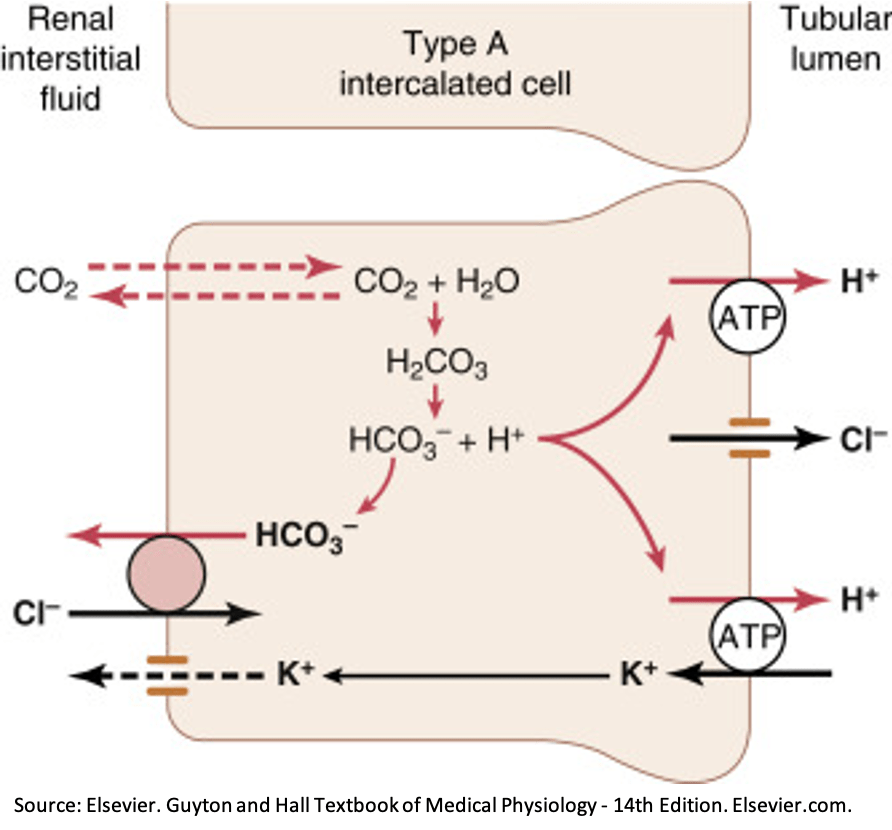
- Volume depletion & increased distal reabsorption. In the distal tubules and collecting ducts, hydrogen ion secretion occurs via the ATP-driven proton pump. In the setting of volume depletion, aldosterone acts on this pump to increase distal hydrogen ion secretion. In addition, aldosterone can indirectly lead to hydrogen ion secretion via the lumen-negative potential differential created by reabsorption of sodium through the ENaC channel.
Correcting volume depletion won’t cure metabolic alkalosis. Chloride will!
While the mechanisms described above provide explanations for the maintenance of metabolic alkalosis after diuretic use, there is evidence suggesting that the correction of volume depletion is not sufficient to correct metabolic alkalosis. For example, in one study, rats were volume expanded with albumin. This did NOT correct the metabolic alkalosis. But chloride salts DID correct the alkalosis, even though the rats remained volume depleted.
Chloride seems to be the key.
One reason that chloride is essential relates to a protein none of us had heard of before 2020: pendrin. Pendrin is a Cl⁻/HCO₃⁻ exchanger in type-B intercalated cells in the cortical collecting duct. When a lot of chloride is delivered to these cells, there is exchange with bicarbonate (see figure below). This allows for bicarbonate excretion. But, if chloride delivery is low, then pendrin cannot excrete bicarbonate and metabolic alkalosis is maintained.

[P = Pendrin]
The key role of pendrin helps explain how we stratify metabolic alkalosis based on urine chloride. Low urine chloride suggests poor delivery to the distal nephron. These patients need chloride. As a result, you will often hear this scenario referred to “chloride depletion alkalosis” and not “contraction alkalosis”.1 In fact, all forms of chloride delivery can improve metabolic alkalosis (e.g., NaCl, KCl, or even HCl).
There are a number of interesting pendrin connections. First, it may be inactivated in cystic fibrosis, leading to metabolic alkalosis when volume depleted.
There is also a genetic disorder (Pendred Syndrome) in which a mutation in the gene encoding pendrin leads to hearing loss (the inner ear contains lots of pendrin). Interestingly, these patients don’t typically have metabolic alkalosis.
Take home points
- Contraction alkalosis refers to a loss of extracellular fluid around a fixed amount of bicarbonate.
- Contraction alkalosis may lead to generation of a metabolic alkalosis but is unlikely to result in its maintenance.
- Under normal conditions, the kidneys are adept at excreting excess bicarbonate.
- Pendrin is a chloride/bicarbonate exchanger that helps to secrete excess bicarbonate.
- With chloride deficiency, pendrin is unable to exchange chloride for bicarbonate and metabolic alkalosis is maintained (“chloride depletion alkalosis”).
Learning Objectives
- Explore how diuretics lead to “contraction alkalosis”.
- Differentiate between how metabolic alkalosis is generated and maintained.
- Introduce the role of pendrin in the resolution of metabolic alkalosis and how it cannot perform this role without adequate chloride delivery.
CME/MOC
We are excited that The Curious Clinicians have partnered with VCU Health Continuing Education to offer continuing education credits for physicians and other healthcare professionals. Visit VCU Health for more information.
Listen to the episode
Credits & Citation
◾️Episode written by Tony Breu
◾️Episode audio by Hannah Abrams
◾️Show notes by Tony Breu
Breu AC, Abrams HR, Cooper AZ. How do diuretics lead to metabolic alkalosis? The Curious Clinicians Podcast. July 22, 2020. https://curiousclinicians.com/2020/07/13/how-do-diuretics-lead-to-metabolic-alkalosis/
Opening image source: https://www.modernhealthcare.com/article/20170526/NEWS/170529910/sodium-bicarbonate-shortage-puts-surgeries-on-hold


Hi, med student here.
I thought diuretics could cause metabolic alkalosis by the induced potassium secretion, since they block NKCC2 or NCC. This means there is more sodium for ENaC and thus K secretion can occur through ROMK.
This leads to hypokalaemia, which cause K to move out of the cells into the extracellular fluid. To counteract the change in charge, protons move in the cells and this reduces the amount of protons in the ECF and thus results in metabolic alkalosis.
Is this not a valid explanation?
LikeLike