Why can’t people with familial fatal insomnia fall asleep?
Have you ever wondered why you feel so awful when you don’t get enough sleep? Well, that’s because it is no exaggeration when we say we need sleep to survive, so much so that we spend approximately ⅓ of our lives sleeping. Insomnia can certainly feel exhausting, overwhelming, and even lonely – while everyone is asleep, there you are – wide awake and unable to fall asleep no matter how much you try. Except, now imagine that other members of your family were plagued with this same condition. And over time, as you remain awake for days to weeks on end, your cognition begins to decline because it has become starved of this essential bodily function. The focus of this episode is a condition called familial fatal insomnia (FFI), a rare and deadly condition that causes progressive neuropsychiatric problems.
We often think of sleep as a state of brain rest and restoration, but it also has significant physiological implications for the whole body. Chronic insomnia has been linked to an increased incidence of infections, as sleep optimizes immune regulation and balances pro- and anti-inflammatory states. Sleep also impacts cardiovascular health. Interestingly, when we look at the fall and spring daylight saving time changes, there appears to be a protective effect on mortality immediately after the fall time change (when people can get an extra hour of sleep) among elderly people, those with dementia, those with chronic respiratory disease, and those with cancer. It’s not a strong association, but people die less often the day after gaining an extra hour of sleep, and the effect persists for 8 weeks after the time change. Unsurprisingly, sleep deprivation also causes cognitive impairment and mood effects.
Sleep is not unique to complex animals such as vertebrates; almost all animals require some form of restorative sleep, including insects. There is evidence that bacteria can engage in cyclical metabolic rest that’s not necessarily sleep, suggesting that living things in general need to rest periodically. An interesting phenomenon is the light-dependent, full-day cycle of photosynthetic cyanobacteria, which oxygenated the early Earth’s atmosphere, beginning about 2.5 billion years ago. As photosynthesizers, they depend on sunlight for their metabolism; at night, they essentially shut down until morning, when they awaken as part of a 24-hour circadian rhythm.
Familial fatal insomnia is a neurodegenerative prion disease that was first reported only 38 years ago, in 1986, in the New England Journal of Medicine. Nine physicians from Italy published the case of a 53-year-old man who had a sharply progressive course of insomnia and signs of dyautonmia, followed by dreamlike mental states, dysarthria, tremor, and myoclonus, which subsequently led him to a coma and death after nine months. What made the case particularly interesting was that other members of his family exhibited similar symptoms and courses of illness. Someone in the family was a physician and observed a pattern of “a peculiar, fatal disorder of sleep” and had been compiling a family history. They were able to trace this syndrome back to the early to mid-1800s, with multiple family members experiencing a neurodegenerative syndrome involving insomnia.
D.T. Max wrote a family profile in the New York Times Magazine in 2001 and later wrote a book about the family. In his Times piece, Max wrote about a man named Giacomo, who lived near Venice, Italy, who suddenly developed insomnia, becoming bedbound and delirious before he died months later at age 45. Over the last two and a half centuries, dozens of family members were similarly affected, and it came to be known as a “family disease” in that part of Italy. As a result, they’ve experienced social isolation, and when Max interviewed a contemporary descendant of Giacomo, she noted that members of the family have trouble getting life insurance.
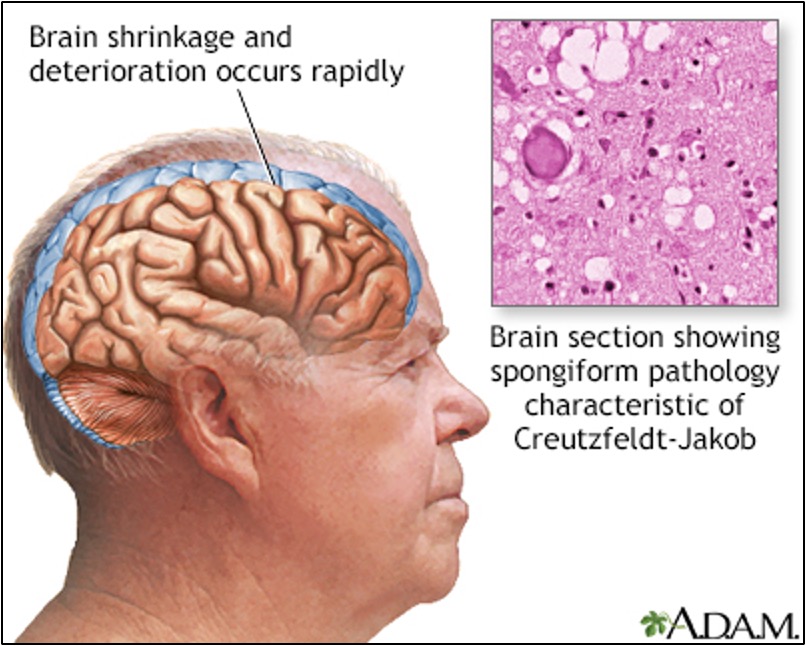
In the 1986 New England Journal case report, they did not know what had caused this inherited disorder, but they noticed some distinct pathological patterns in the patient’s brain as well as the brain of his sister, who had died of the same disease. The authors found neuronal death in specific thalamic regions in both brains. They wondered whether this could be a form of Creutzfeldt-Jakob Disease (CJD), but felt that this Italian family’s presentation did not align with what is typically seen in CJD.
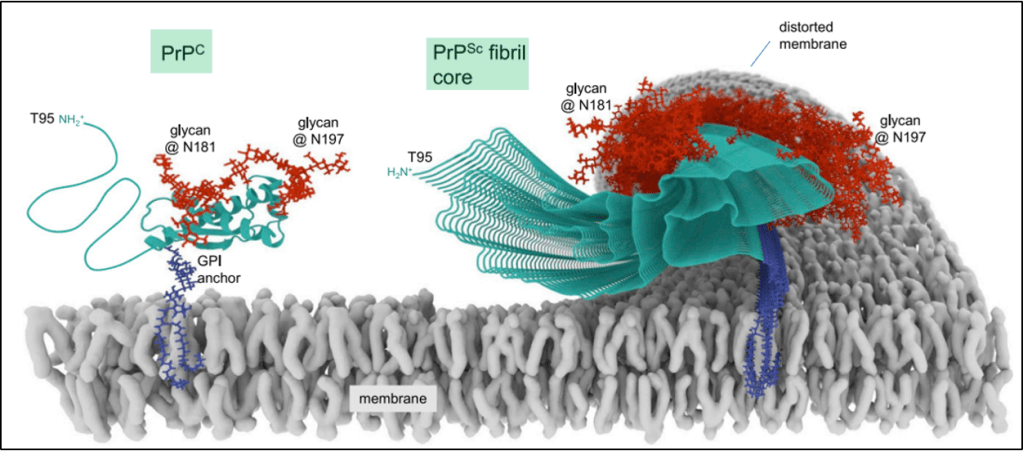
FFI is also a prion disease, similar to CJD. Inherited mutations in the prion gene PRNP cause FFI. There are sporadic cases too, where a PRNP mutation arises de novo. It’s separate from CJD, scrapie, or mad cow disease – the so-called transmissible spongiform encephalopathies. There actually is a physiologic version of prion proteins known as PrP. PrP is a type of protein that we all have that’s bound to cell membranes and transports copper, and it is necessary for the function of neurons in particular.
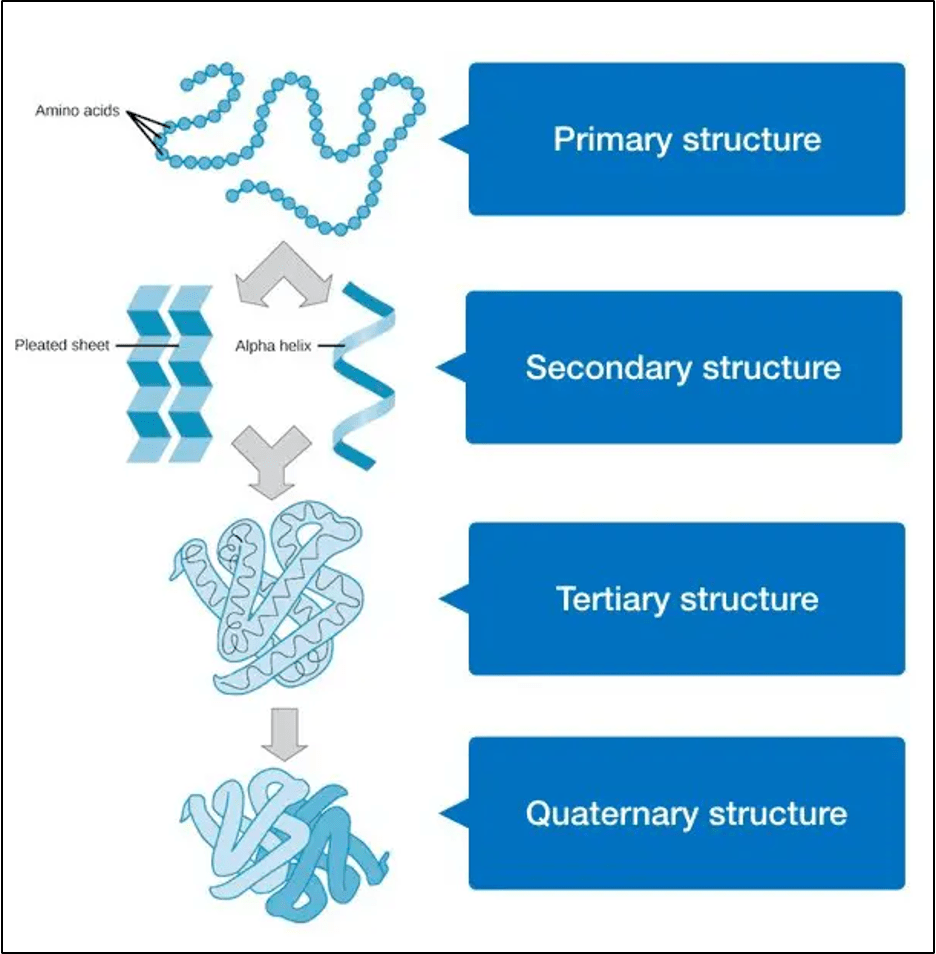
In the case of pathologic prions, a change in the folding pattern leads to disease. Proteins fold on themselves. The primary structure of proteins is the amino acid sequence that determines how the protein folds. This is followed by secondary folding, which involves two types of patterns called motifs: beta-sheets or alpha-helices. With these neurotoxic prion proteins, either a genetic mutation or an acquired infectious prion protein initiates a cascade of conversion of alpha-helices to beta–sheets. The proteins then start to stack and roll into themselves as their secondary structures all snap into alignment with each other.
With neurons, this cascade leads to neurotoxicity and neurodegenerative symptoms. Acquired prion diseases can be caught by eating infected nervous tissue, such as beef from affected animals, which can cause mad cow disease, also called variant Creutzfeldt-Jakob disease. Exposure to brain matter or cerebrospinal fluid of an affected person, whether from contaminated surgical instruments or from body fluid exposure after a lumbar puncture, can also cause it. These occurrences are exceedingly rare due to safety regulations in the food supply and healthcare facilities.
So, why are people with FFI unable to sleep? This actually has to do with the location of where these abnormal prion proteins are forming. As noted earlier, in the 1986 case report, the authors found thalamic degeneration at autopsy. This anatomic pattern has been confirmed with subsequent case series as well, with the consistent finding that familial fatal insomnia is ultimately a thalamic disorder. Below is a PET scan example from the brain of someone with FFI. There is no metabolism in the thalamus; that part of the brain shuts down.
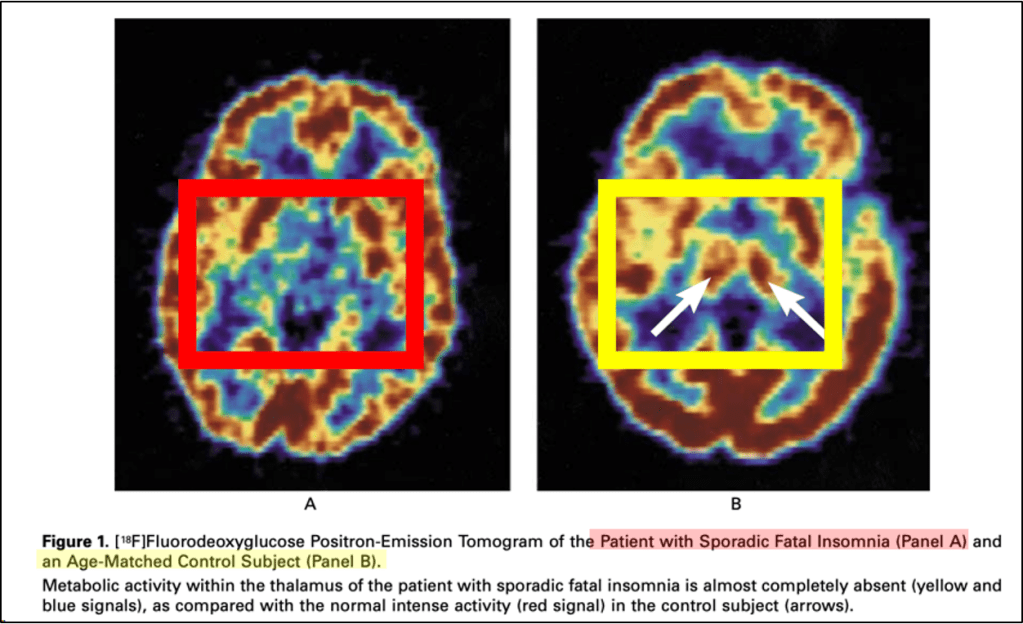
Since the thalamus is a primary regulator of the sleep-wake cycle, from wakefulness to non-REM to REM sleep, its localization is reasonable. The thalamus is hyperconnected to the rest of the brain, like an old-school phone switchboard operator, and one of the many things it regulates is sleep. It is not fully understood why the thalamus is uniquely susceptible in individuals with a PRNP gene mutation, compared with other brain regions that carry the same mutation. There’s two plausible mechanistic theories that we can briefly review. The first was that there might be specific receptors for these abnormal prions that are present only in the thalamus. One proposed candidate is the limbic associated membrane protein or LAMP, which is a type of cell membrane receptor that only exists on neurons in parts of the brain within or closely associated with the thalamus. Another theory is that different degrees of prion glycosylation inside the thalamus could affect how these prions function.
Currently, FFI has no cure but doxycycline is being tested to see if it will slow down the disease or prevent it from arising altogether. Doxycycline has two effects that may be beneficial in FFI: it blocks the formation of abnormal prion protein and increases its proteolytic degradation. A 2021 paper out of Italy published in the journal Neurobiology of Disease found that giving prophylactic doxycycline to mice in an in vivo familial fatal insomnia model did minimize memory and sleep issues that the animals would have otherwise developed, however they still experienced fatal neurodegeneration. From a clinical standpoint, a 10-year preventive doxycycline trial testing in families affected by the disease concluded recently, but the results have not yet been published.
Additionally, a couple of avenues have been pursued, leveraging the observation that a very small percentage of people are what’s called “PRNP heterozygous”, having only one copy of the PRNP gene, and so make less than normal amounts of prion protein. They seem to suffer no ill effects. One approach is to silence the abnormal prion gene using an epigenetic editor. The name for this is CHARM (Coupled Histone tail for Autoinhibition Release of Methyltransferase), as reported in a 2024 paper in the journal Science from Harvard and MIT that tested this technology in a mouse model. The researchers used a viral vector to deliver CHARM to the mice’s brains, thereby silencing prion genes by inducing DNA methylation at those sites. This can offer therapeutic avenues for other prion diseases besides just familial fatal insomnia. Another method is to edit the PRNP gene itself to definitively silence it in vivo using CRISPR technology, which the same group of researchers found could decrease prion protein expression in PRNP-mutated mouse brains by 50% and extend survival by the same margin.
The author list of the two papers cited above, which are significant advances in prion research, includes Dr. Sonia Vallabh as the second-to-last author. She is a neuroscientist at Harvard Medical School who also found out that she inherited a PRNP mutation after her mother died of familial fatal insomnia in 2010. She and her husband, Dr. Eric Minikel, run a lab studying prion disease therapeutics at the Broad Institute in Boston. Here is a link to a 2025 Harvard Gazette article that highlights their incredible story.
Take Home Points
- Familial fatal insomnia is a neurodegenerative prion disease that leads to, among many clinical symptoms, progressive insomnia. Misfolded prion proteins accumulate in the thalamus
- Thalamic prion accumulation then causes disruption in the sleep-wake cycle and an inability to fall asleep
- It is not fully understood why this thalamic specificity exists, but it may result from different glycosylation of prions in the thalamus or because of prion receptors that only exist in that part of the brain.
- There are fascinating potential therapies being studied and under development, including prophylactic doxycycline, epigenetic prion gene silencing, and gene editing technologies using CRISPR
CME/MOC
Click here to obtain AMA PRA Category 1 Credits™ (0.5 hours), Non-Physician Attendance (0.5 hours), or ABIM MOC Part 2 (0.5 hours).
As of January 1, 2024, VCU Health Continuing Education will charge a CME credit claim fee of $10.00 for new episodes. This credit claim fee will help to cover the costs of operational services, electronic reporting (if applicable), and real-time customer service support. Episodes prior to January 1, 2024, will remain free. Due to system constraints, VCU Health Continuing Education cannot offer subscription services at this time but hopes to do so in the future.
Credits & Suggested Citation
◾️Episode written by Avi Cooper
◾️Show notes written by Avi Cooper and Millennium Manna
◾️Audio edited by Clair Morgan of nodderly.com
Cooper AZ, Abrams HR, Breu AC, Buonomo G, Manna, M. No Sleep for the Weary. The Curious Clinicians Podcast. December 10th, 2025.
Image Credit: Pexels.com
Original tweetorial source: https://x.com/AvrahamCooperMD/status/1837872885018341658?s=20

