Why don’t interstitial lung disease patients typically get increased pCO₂?
Early on in medical training, you learn to separate a patient’s oxygen from their carbon dioxide. A COPD patient can be heard wheezing from across the ward with a pCO₂ of 80mmHg and actually have a stable O₂ saturation, while their neighbor with interstitial lung disease will be barely maintaining their oxygen on six liters and have a pCO₂ of 40mmHg. The thinking, simply put, is that O₂ is about air “in” and pCO₂ is about air “out.” That makes sense, until you think about what is actually happening in the lung. Someone with ILD is hypoxemic because their interstitium is so fibrotic and thick that diffusion of O₂ into the pulmonary capillaries is impaired. But just as O₂ needs to diffuse into the blood to be absorbed, CO₂ needs to diffuse out of it to be exhaled. So why doesn’t that patient with ILD also have a high pCO₂?
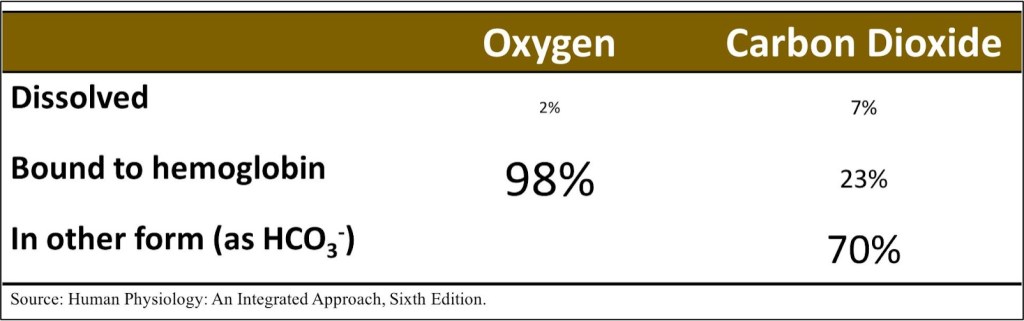
The answer lies in that dreaded term: The gas laws. Don’t worry, this will all make sense by the end. We need to start with Henry’s Law, named after the 18th century English chemist and physician William Henry. It states that the concentration of a dissolved gas is proportional to its partial pressure and to the solubility coefficient. The higher the coefficient, the more soluble it is. The solubility coefficients of O₂ and CO₂ are quite different: O₂ has a coefficient of 0.024, while CO₂ has one of 0.57. This makes CO₂ about 24 times as soluble as O₂.
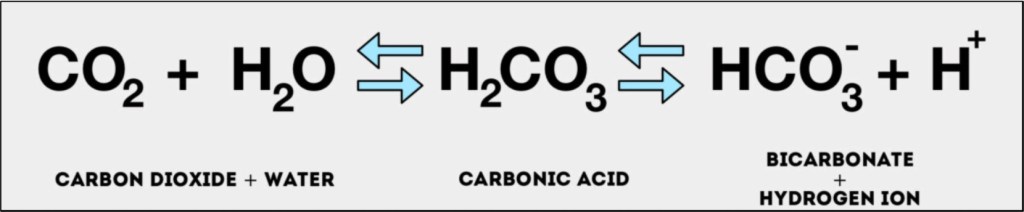
Why such a big difference? Two reasons. First, our blood is mostly water, and H2O is a highly polar molecule. Although CO₂ is overall nonpolar, its polar C=O bonds allow favorable interactions with water. O₂, by contrast, is completely nonpolar and does not interact well with H2O. Second, CO₂ doesn’t just interact with water, it reacts with it, forming carbonic acid, which in turn is converted to bicarbonate and a hydrogen ion. As the CO₂ is converted into carbonic acid, chemical equilibrium then favors more CO₂ dissolving. This is a demonstration of Le Chatelier’s principle, named for the 19th-century French chemist Henry Louise Le Chatelier. This reaction explains why only about 7% of the CO₂ carried in the blood is actually dissolved CO₂ – the vast majority is carried as bicarbonate.
Ok, so this all explains pretty well why CO₂ is so much more soluble in the blood than O₂ is. But in the end, we’re breathing out CO₂, not bicarbonate, and these laws don’t seem to be the passport that lets CO₂ into the United States of Alveoli while O₂ is languishing in the customs line on the other side. For that, we turn to Fick’s Law of Diffusion, a name many physicians may only know from cardiac catheterization. Named for Adolf Fick, a German-born physician and physiologist, this law states states that the rate at which a gas moves across a membrane is proportional to the solubility of the gas, the surface area of the membrane, and the difference in partial pressures across the membrane, and inversely proportional to the membrane’s thickness and the molecular weight of the gas. In other words, the smaller and more soluble gas is, it moves more easily across a given membrane. CO₂ is, obviously, a bit heavier than O₂, but it is much more soluble, and on balance already moves across the alveolar membrane more easily.
We can then return to Le Chatelier’s principle that if a system at equilibrium is disturbed, the system shifts to counteract that change. On its journey from the tissues to the lungs, an equilibrium exists between the reactions that convert CO₂ to carbonic acid and that in turn to bicarbonate and a hydrogen ion. When the blood reaches the alveolar membrane, the dissolved CO₂ starts to slip across it, which then favors the production of more CO₂ in the blood to try and restore the previous equilibrium, and that then escapes, and the cycle continues. Since Fick’s Law says diffusion is also proportional to the partial pressure difference across the membrane, as more CO₂ is created in the blood, the pressure gradient favors it passing into the alveoli.
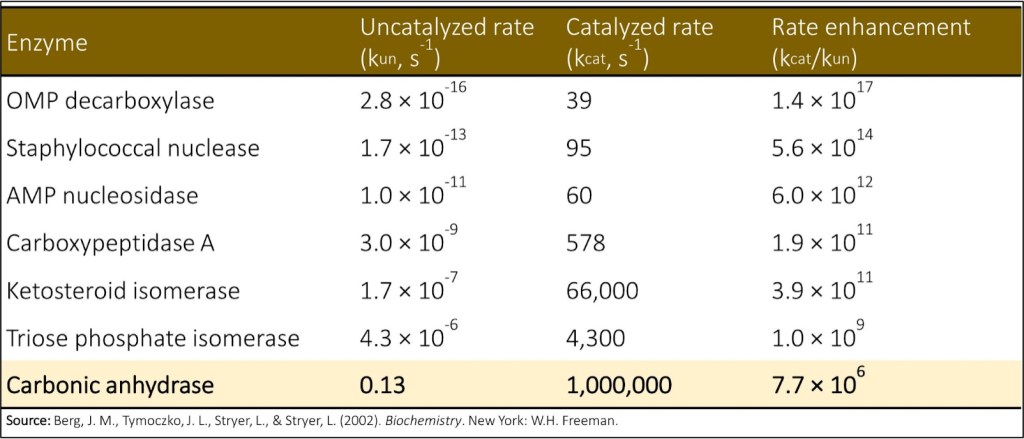
So there’s our answer! Almost. One thing you need to know about that CO₂/carbonic acid system is that it’s very, very slow, at least in the biochemical sense. It takes several seconds to react to changes and reach a new equilibrium. Given that the transit time of a red blood cell across an alveolus is under one second, there’s simply not enough time for that crafty little use of Le Chatelier’s to have any real effect. And then comes carbonic anhydrase. This enzyme, which is in red blood cells, takes the system from one reaction every five seconds to a million every one second. When venous blood first reaches tissue, carbonic anhydrase ensures that CO₂ is rapidly converted to carbonic acid, which creates a positive gradient for continued CO₂ diffusion out of cells. At the end of the journey, it reverses roles and allows for CO₂ to be continually produced so it can diffuse across the membrane. Carbonic anhydrase is so vital to these processes that acetazolamide, a carbonic anhydrase inhibitor used to treat idiopathic intracranial hypertension, edema and many other conditions, can actually cause severe respiratory acidosis in patients with COPD.
The strongest evidence, in a sense, for how unaffected CO₂ is by things like interstitial thickening or alveolar filling is the equation for its partial pressure in arterial blood. It’s proportional to tissue CO₂ production and inversely proportional to alveolar ventilation. As long as air is entering and leaving the alveoli, CO₂ will diffuse across the membrane. That’s why our patient with COPD, who can’t get enough air out, has a high pCO₂, but our ILD patient, who has enough air in and out of the alveoli and just can’t get the O₂ across the membrane, doesn’t have to add hypercapnia to their list of problems.
Take Home Points
- We don’t typically see hypercapnea in interstitial lung disease, even though we see hypoxemia.
- The reasons for this difference are that carbon dioxide is about 24 times more soluble than oxygen, plus the gradient for alveolar diffusion is maintained by the ultrafast carbonic anhydrase reaction.
- This allows for continued CO₂ diffusion across a thickened interstitium
Sponsor
This episode was sponsored by FIGS. FIGS is offering 15% off your first purchase. Just go to wearfigs.com and use the code FIGSRX at checkout
Listen to the episode!
https://directory.libsyn.com/episode/index/id/40790570
CME/MOC
Click here to obtain AMA PRA Category 1 Credits™ (0.5 hours), Non-Physician Attendance (0.5 hours), or ABIM MOC Part 2 (0.5 hours).
As of January 1, 2024, VCU Health Continuing Education will charge a CME credit claim fee of $10.00 for new episodes. This credit claim fee will help to cover the costs of operational services, electronic reporting (if applicable), and real-time customer service support. Episodes prior to January 1, 2024, will remain free. Due to system constraints, VCU Health Continuing Education cannot offer subscription services at this time but hopes to do so in the future.
Credits & Suggested Citation
◾️Episode written by Tony Breu
◾️Show notes written by Tony Breu and Giancarlo Buonomo
◾️Audio edited by Clair Morgan of nodderly.com
Breu AC, Abrams HR, Cooper AZ, Buonomo G, Manna, M. One of the Fast Reactions in the Body. The Curious Clinicians Podcast. April 1st, 2026.
Image Credit : Adobe