Why does an autoimmune antibody stimulate the thyroid in Graves’ disease?
When we think of antibodies, such as monoclonal antibodies that target specific diseases, we often think of them as blocking receptor-ligand interactions or binding to pathogens to neutralize them. Yet in Graves’ disease, we actually find the opposite of what we would normally expect: an antibody actually activates the receptor rather than inhibit it.
Although Graves’ disease was first described in 1786, the condition was actually initially described by a physician from England, Caleb Parry. He gathered a case series of patients during his clinical practice in Somerset, and these patients had clinical symptoms that fit with the disease we now call Graves’ disease. According to Parry’s description:
“The part swelled was the thyroid gland; the eyes were protruded from their sockets, and the countenance exhibited an appearance of agitation and distress, especially on any muscular exertion, which I have rarely seen equalled.”
Parry’s descriptions from the late 1700s were published posthumously in 1825 by his son, Charles Parry. Parry was a very curious and observant person, as evidenced by his extensive writings on rabies, which he called ‘canine hydrophobia.’ While Parry described Graves’ first, the Irish surgeon Robert Graves published a case report in 1835 about a man with enlarged eyes, difficulty shutting them, and an enlarged thyroid. Graves himself was a remarkable and fascinating individual, ahead of his time on many topics, including the adoption of the newly invented stethoscope, the use of the second hand on a watch to measure heart rate, and feeding febrile patients, which was thought to be harmful at the time. Several years later, Armand Trousseau suggested naming this syndrome “Graves’ disease,” and that stuck, although William Osler recognized that Parry had been overlooked and should have had priority in naming.
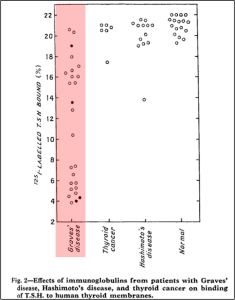
Despite Parry’s clinical observations in the late 1700s, it wasn’t until 2 centuries later, in the 1950s, that the pathophysiology of Graves’ disease began to be unpacked. Most of the work was done by two researchers named Adams and Purves. Their work was based on the observation that injecting thyroid-stimulating hormone (TSH) into guinea pigs led to the thyroid gland producing thyroid hormone. The effects of TSH would peak about 3 hours after administration. However, when they injected these guinea pigs with serum samples from patients with hyperthyroidism or thyrotoxicosis, they noticed that something in the samples stimulated the thyroid gland, with effects peaking at about 24 hours. Though much more delayed than TSH, they also noticed it lasted longer in the system. They initially called this non-TSH thyroid-stimulating substance “long-acting thyroid stimulator.” Further work then identified that it was an antibody. Interestingly, at the time they made this discovery, the TSH receptor had not yet been discovered. The TSH receptor was discovered in 1973, and the following year, this thyroid-stimulating factor was shown to be an antibody that binds the TSH receptor very tightly and was labelled the thyrotropin receptor antibody.
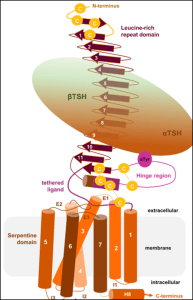
So, why is it that antibodies activate rather than inhibit their receptor in Graves’? This is because the TSH receptor has a repeating domain that is rich in leucine that extends from the transmembrane portion of the receptor. This leucine-rich repeat in the schema looks like the head of a lacrosse stick, the part that catches the ball. The TSH receptor binds this head, activating G protein-coupled signaling in thyrocytes. A study published in the journal Thyroid by a group of biochemists from the United Kingdom in 2007 used X-ray crystallography to examine how the TSH receptor and a thyroid-stimulating antibody in Graves’ disease interact at the molecular level. They found that the thyroid-stimulating antibody binds to the TSH receptor at essentially the same location and configuration as TSH itself, even though they do not look alike. They also noted that the thyroid-stimulating antibody binds very tightly and dissociates slowly, which explains the significantly longer TSH receptor activation time compared with TSH itself.
While this antibody can bind the receptor, the results are not uniform because multiple types of anti-TSH receptor antibodies are produced in Graves’ disease. Some stimulate the receptor and activate the thyroid; some are neutral in their effects; and some actually block TSH from binding but don’t stimulate the thyroid, as we typically associate antibodies with antagonism. These would be considered thyroid-blocking antibodies instead of thyroid-stimulating antibodies. So any given patient with autoimmune thyroid disease could potentially produce a combination of these different antibody types.
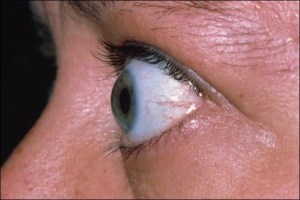
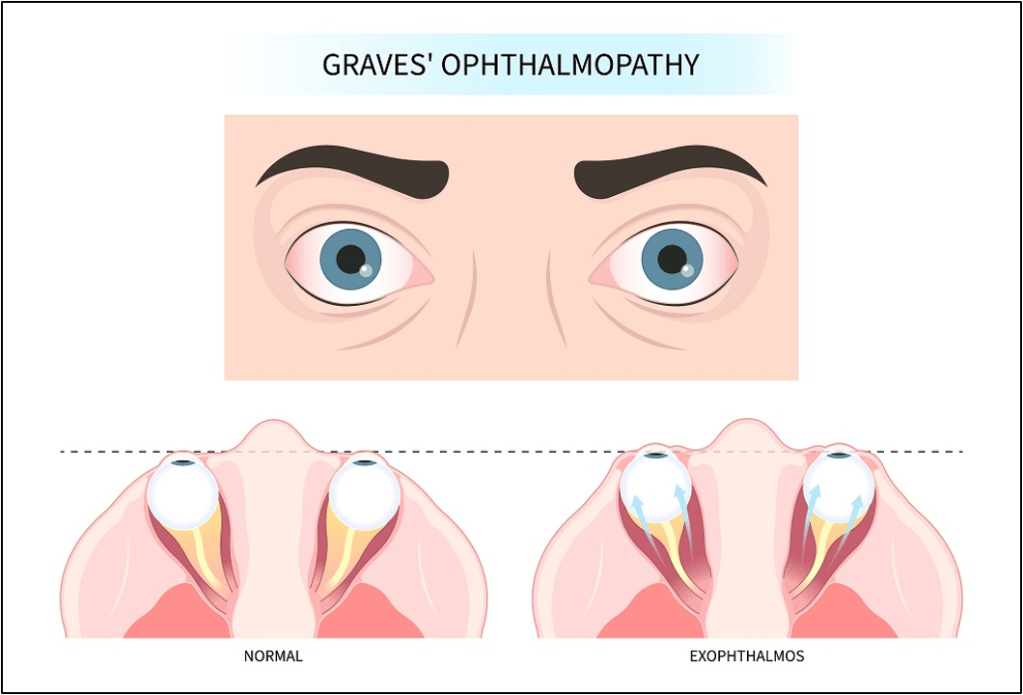
Graves’ ophthalmopathy, also known as Graves’ orbitopathy, is due to edema and fibrosis of the extraocular muscles and tissues of the eye orbit. This clinical phenomenon occurs because TSH receptors are also found on the cell surfaces of orbital fibroblasts. TSH receptor antibodies stimulate orbital fibroblasts to produce glycosaminoglycans and collagen in the tissues around the eyes, and induce an inflammatory reaction in the orbits themselves, causing edema and inflammation of the extraocular muscles. The result is what we see clinically: protrusion of the eye, known as exophthalmos.
Take Home Points
- Graves’ disease results from thyroid-stimulating antibodies binding to the TSH receptor in essentially the exact same location and configuration as TSH itself does, even though they don’t look anything alike.
- Patients with autoimmune thyroid disease also can produce that block the TSH receptor instead of stimulating it.
- Graves’ ophthalmopathy results from activation of orbital fibroblasts that also express TSH receptor
CME/MOC
Click here to obtain AMA PRA Category 1 Credits™ (0.5 hours), Non-Physician Attendance (0.5 hours), or ABIM MOC Part 2 (0.5 hours).
As of January 1, 2024, VCU Health Continuing Education will charge a CME credit claim fee of $10.00 for new episodes. This credit claim fee will help to cover the costs of operational services, electronic reporting (if applicable), and real-time customer service support. Episodes prior to January 1, 2024, will remain free. Due to system constraints, VCU Health Continuing Education cannot offer subscription services at this time but hopes to do so in the future.
Credits & Suggested Citation
◾️Episode written by Avi Cooper
◾️Show notes written by Avi Cooper and Millennium Manna
◾️Audio edited by Clair Morgan of nodderly.com
Cooper AZ, Abrams HR, Breu AC, Buonomo G, Manna, M. Why-roid. The Curious Clinicians Podcast. March 19, 2026.
Image Credit: Endocrine Society
Original tweetorial: